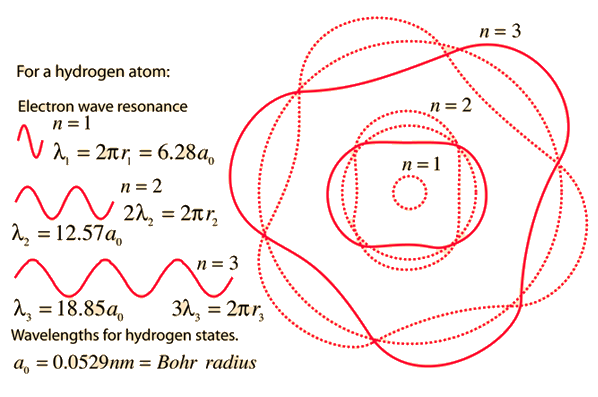
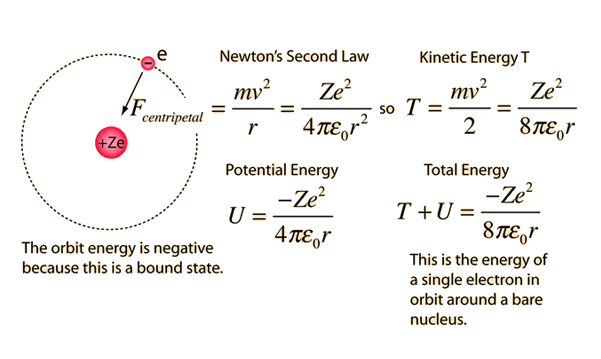
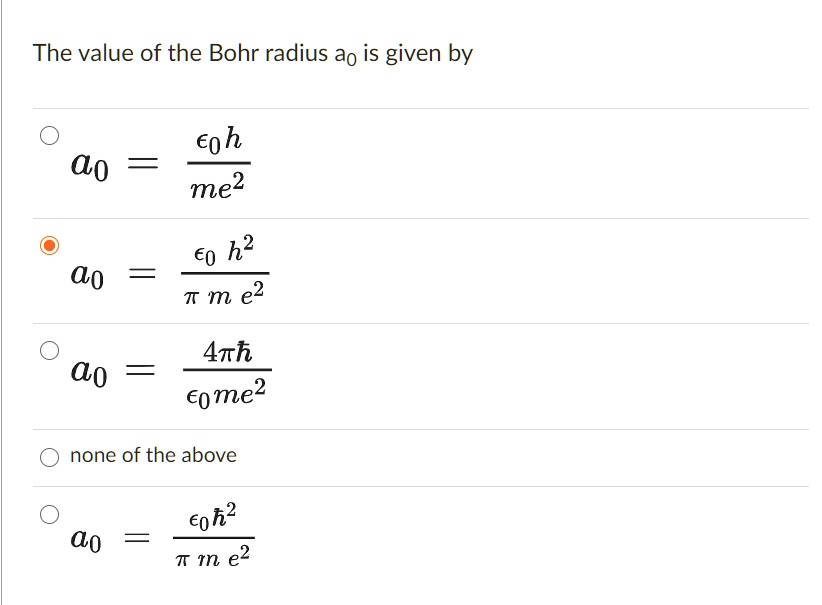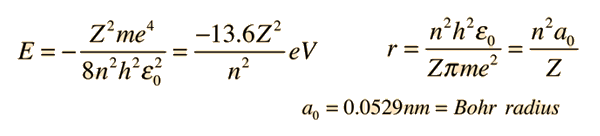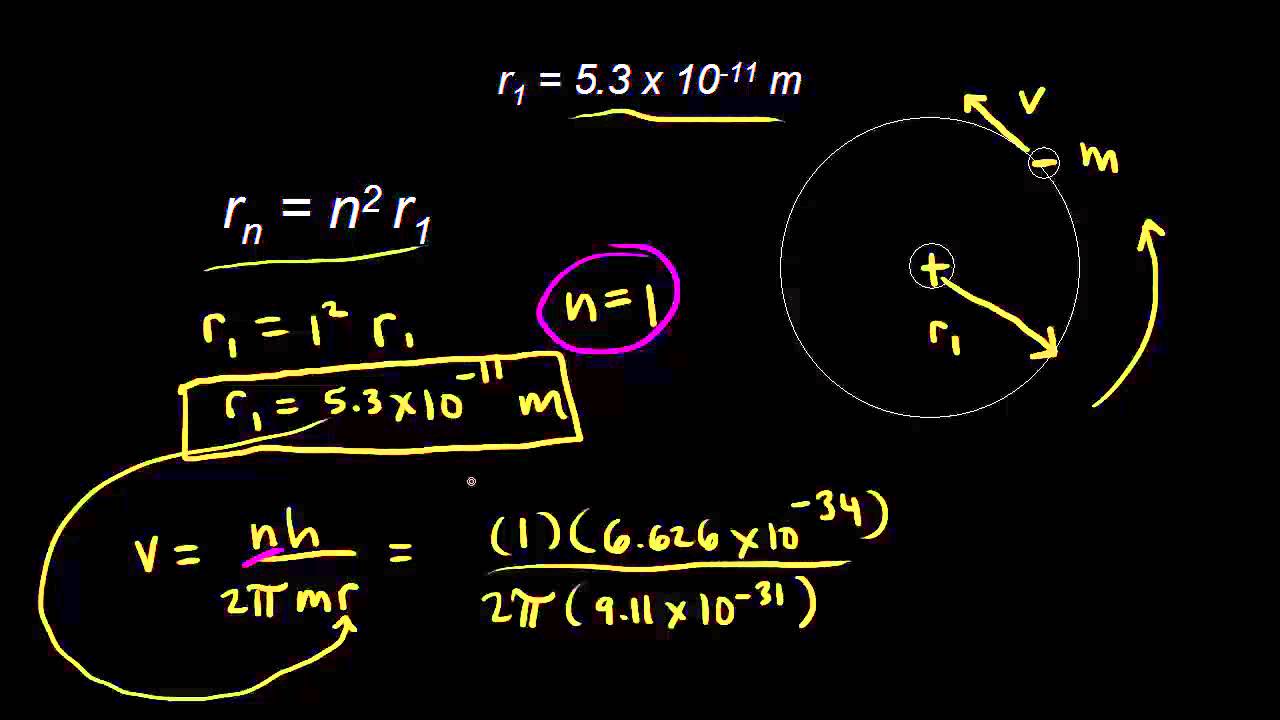1. The first Bohr's radius (for electron in hydrogen atom in the ground state): 2. The ground energy level in hydrogen atom: - ppt video online download

In the ground state of hydrogen atom, its Bohr radius is 5.3xx10^(-11)m. The atom is excited such that the radius becomes 21.2xx10^(-11)m. Find the value of principal quantum number and total energy

In the Bohr's model of hydrogen - like atom the force between the nucleus and the electron is modified as F = e^24piε0 ( 1r^2 + betar^3 ) , where beta is
What will be the radius of the third orbit, if the radius of the first orbit of an electron is r in Bohr's model of hydrogen atom? - Quora

Radius of nth Bohr's orbit-Equation -Shortcut formula-Solved problems-IIT JEE NEET GATE Adichemistry - YouTube
Derive a formula for radius of the stable orbit of hydrogen atom on the basis of Bohr model. Prove that in hydrogen - Sarthaks eConnect | Largest Online Education Community

Show all steps of the derivation: Show that the most probable radius for an electron described by the function R_10 is the Bohr radius, a_0. | Homework.Study.com